An Auger-Emitting Radioconjugate for Triple-Negative Breast Cancer

Cellectar Biosciences enrolls first patient in Phase 1b of CLR 125, an iodine-125 Auger emitter targeting refractory TNBC.

Cellectar Biosciences enrolls first patient in Phase 1b of CLR 125, an iodine-125 Auger emitter targeting refractory TNBC.

Grace Science earns RMAT designation for GS-100, the only gene therapy in development for ultra-rare NGLY1 deficiency.
ImmuVia's bispecific IMV-M targets MUC16 first, then triggers DR5-mediated apoptosis in ovarian and pancreatic tumors.
GlycoNex receives PMDA approval to start first-in-human trials of GNX1021, a glycan-targeting ADC for GI cancers.

TOBY Inc receives FDA Breakthrough Device Designation for a urine-based multi-cancer early detection test.
Dyve Biosciences reports preclinical data for DYV800, a transdermal bicarbonate therapy that resets tumor pH to restore immune function.
Hinge Bio doses first patient with HB2198, a multispecific antibody targeting CD19 and CD20 for autoimmune disease.
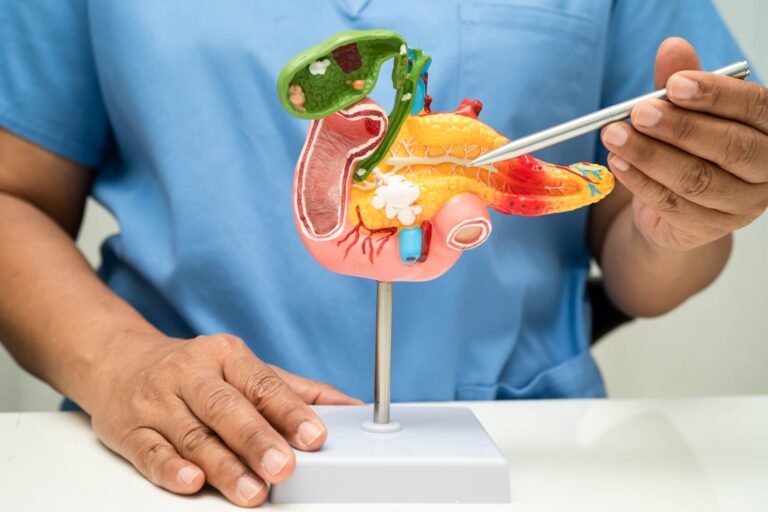
Actuate Therapeutics publishes Phase 2 elraglusib data in Nature Medicine, doubling one-year survival in pancreatic cancer.

Novo Nordisk partners with OpenAI as AI drug discovery deal flow surpasses $5B in five months.
PannTheraPi files for Phase 2a trial of first-in-class pannexin-1 inhibitor for drug-resistant childhood epilepsy.