Ten days ago, Vivatides raised $54 million to engineer RNA therapeutics that route around the liver.
At AACR 2026 this weekend, Nanobiotix showed up with preclinical data on a completely different approach to the same problem.
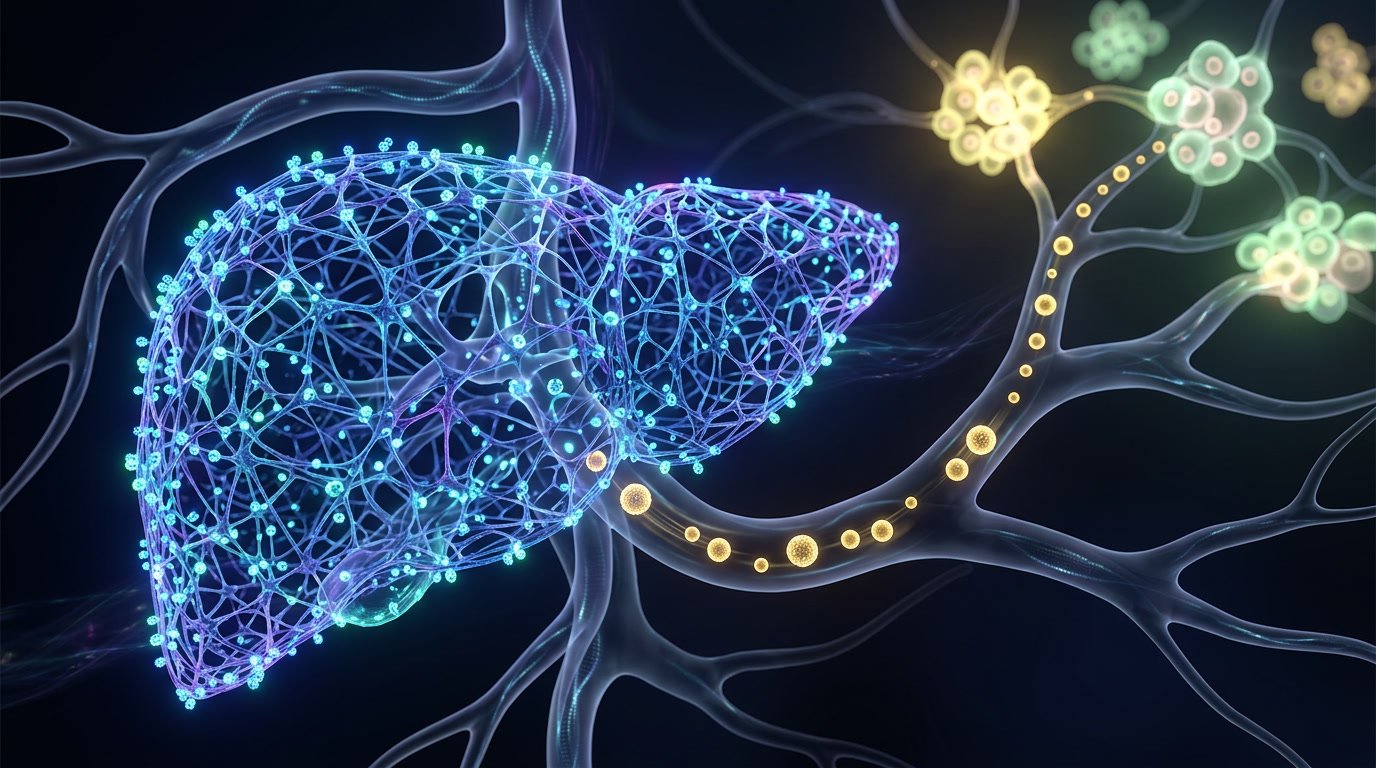
Don’t engineer a delivery vehicle that avoids the liver…distract the liver first, then dose the regular LNP.
The platform is called Nanoprimer (the Curadigm business line, if you’ve been following Nanobiotix for a while).
It’s a decoy nanoparticle designed to temporarily occupy the mononuclear phagocyte system, which is the hepatic clearance pathway that chews up roughly 90% of systemically dosed LNPs before they reach any other tissue.
Pre-treat with Nanoprimer. The liver gets busy mopping up the decoy. Then you dose your actual LNP payload and a lot more of it survives to reach extrahepatic tissue.
That’s the idea anyway.
Two approaches to the same problem
Vivatides approach
Redesign the payload
Engineer RNA delivery vehicles with ligands that target non-liver tissue. Skip the liver entirely.
Nanobiotix approach
Saturate the liver
Pre-dose a decoy that temporarily occupies hepatic clearance, then dose the regular LNP.
In mouse models, Nanoprimer pre-treatment improved systemic bioavailability of LNP-delivered DNA immunotherapy constructs and reduced liver accumulation. It also attenuated toxicity and inflammation, which they attributed to reduced cGAS-STING pathway activation (this is a known problem with LNP-delivered nucleic acids, where the innate immune system reads your therapy as a viral infection and starts pumping out type I interferons … toxic. No good. Very bad.).
This is preclinical, presented as a poster – so, grain of salt.
However, I’d say that the meaningful read on this data isn’t the magnitude of the effect. It’s that a company with an existing Phase 3 program (NBTXR3 in head and neck cancer, partnered with J&J) has a second platform that solves a completely different problem in a crowded adjacent field.
The Biotech Voyager
Early-stage biotech signals, personalized.
The signals that matter to you, contextualized and written directly to you, so you cut through the noise and immediately understand why it matters.
The two approaches aren’t mutually exclusive either.
Vivatides, Sarepta, Atrium and the rest of the extrahepatic delivery field are building ligand-targeted delivery vehicles.
Those work by being smarter than liver clearance. Nanobiotix is building something that makes liver clearance briefly stupider. You could imagine using both.
What’s novel about the Nanoprimer is the business model. They want to be the delivery layer that every LNP-based company uses. Four new patents filed in November 2025. Multiple material transfer agreements in place. They’re positioning this as platform infrastructure.
Whether that works depends on whether the data holds up in humans and whether LNP-based companies are willing to add a pre-dose step to their clinical protocols.
Patients will need to be dosed twice.
Which makes the trial design more complex.
There’s a real ask to any partner.
But if the extrahepatic delivery problem is the bottleneck the entire RNA therapeutics field has been stuck on for a decade, and if this decoy approach works cleanly in humans, Nanobiotix has a second franchise on its hands.
Keep watching for data readouts in 2026. This is the kind of thing that goes from curiosity to infrastructure once the first partnership gets announced.